RESEARCH:
Transcription Stress & Genome stability:
LINKS:
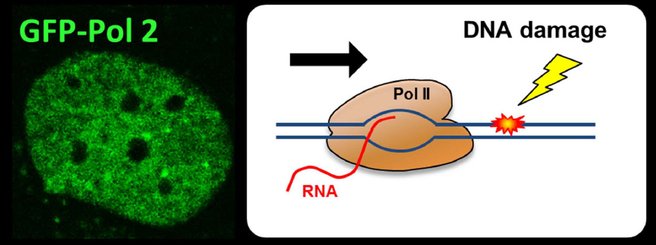
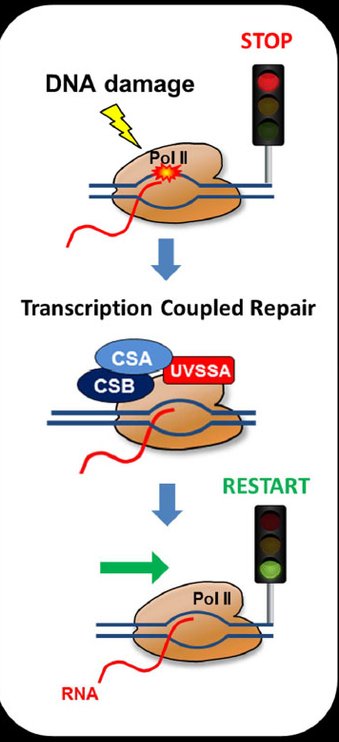
The eukaryotic genome is transcribed by RNA polymerase 2 (Pol 2). Pol 2 mediated transcription is a tightly controlled process to ensure correct temporal and spatial regulation of gene expression, which is crucial for proper cell function. However, the DNA template that is transcribed by Pol 2 is compromised on a daily basis by numerous types of DNA-damaging factors. DNA damage can block or strongly impede Pol 2 mediated transcription. If these transcription-blocking DNA lesions (TBLs) are not properly resolved, this will severely disrupt cellular homeostasis due to the disturbed synthesis of new RNA molecules. In addition, TBLs may result in genome instability, severe cellular dysfunction or premature cell death, which finally may result in DNA damage-induced accelerated aging. Cells counteract these harmful effects by specifically removing TBLs by transcription-coupled repair (TCR), thereby safeguarding transcription. The severe developmental, neurological and premature aging features observed in patients with inherited TCR defects underscore the biological relevance of TCR and show the involvement of TBLs in the aging process.
The main research focus of my lab is to investigate the precise regulation and impact of TCR. Over the years, my lab has identified several factors that play important roles in this repair pathway. Although these studies resulted in new insights in the molecular mechanism of the TCR reaction itself, thus far surprisingly little is known about the regulation of transcription or what exactly happens with Pol 2 following the induction of transcription-blocking DNA damage. For example, the exact mechanisms how transcription is inhibited upon DNA damage, or how it is restarted following repair of the TBLs remains largely enigmatic. We use a combination of innovative live-cell imaging approaches with advanced proteomics and genomics approaches to identify new factors involved in the regulation of this pathway and to obtain a better understanding of the molecular mechanisms how cells cope with transcription blocking DNA damage.